Microbial interactions with carbon nanotube-polymer nanocomposites
Manufacturers of plastics and other polymers use a variety of additives in order to improve the properties and expand the commercial applications of their products. Carbon nanotubes (CNTs) are one such additive. When these long, thin graphene cylinders are embedded in plastics, the resulting CNT-polymer nanocomposites (CNT-PNCs) have enhanced tensile strength, electrical conductivity, and thermal stability compared to the same plastics without CNTs. The properties of CNT-PNCs are important not only in terms of their commercial value, but also in light of the potential environmental impacts of nanocomposites. Questions about how engineered nanomaterials like CNT-PNCs may behave in the environment abound.
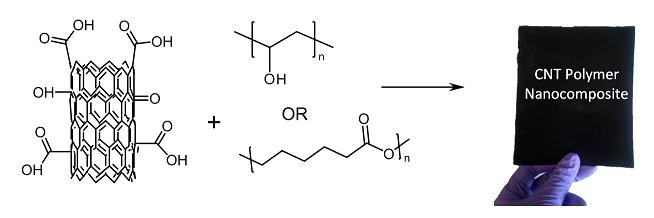
Once a product containing CNT-PNCs reaches the end of its useful life and is discarded, its long-term fate depends on where it ends up and how it breaks down. Biological, physical, and chemical processes in landfills, soils, and surface waters will affect how fast the product degrades and whether or not the CNTs are released into the environment (and to what effect). To improve understanding of the potential impacts of CNT-PNCs on microbial populations and to help inform safe design of CNT-PNC products, the Bouwer group is collaborating with Howard Fairbrother’s group from the JHU Department of Chemistry to explore the effects of the added nanotubes on interactions between CNT-PNCs and bacteria.

One joint study focuses on how CNTs can decrease a plastic’s susceptibility to biodegradation. Starting with an easily biodegradable polymer known as PCL (poly-ε-caprolactone), we add CNTs to form a nanocomposite with a range of nanotube concentrations so that we can quantify how the CNT loading affects the biodegradability of the PCL under various conditions. After initial testing with a model laboratory organism, Pseudomonas aeruginosa, we are branching out to consider other organisms and more environmentally relevant conditions, including aerobic biodegradation by a mixed culture of wastewater microorganisms and anaerobic biodegradation by microbes from landfill leachate.
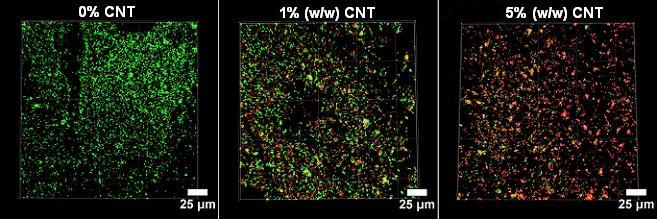
A second joint study focuses on the antimicrobial surface properties of CNT-PNCs made from a polymer called PVOH (polyvinyl alcohol). Using confocal laser scanning microscopy (CLSM) to visualize both living and dead bacterial cells, our researchers can study how the CNT concentration in the nanocomposite affects the viability of microbes that are exposed to the surface of the plastic for short periods of time. In environmental contexts, bacteria often colonize surfaces as biofilms, so Bouwer group researchers are also studying the longer-term effects of exposure to CNT-PNC surfaces on biofilm formation in a drip-flow reactor.